Solar cell is a device or a structure that converts the solar energy i.e. the energy obtained from the sun, directly into the electrical energy. The basic principle behind the function of solar cell is based on photovoltaic effect.
Solar cell is also termed as photo galvanic cell. The electricity supplied by the solar cell is DC electricity / current which is same like provided by batteries but a little bit different in the sense the battery is providing constant voltage.
In another way of defining the solar cell it is a solid state electrical device that converts energy of light directly into electricity by Photoelectric Effect.
Construction of Solar Cell
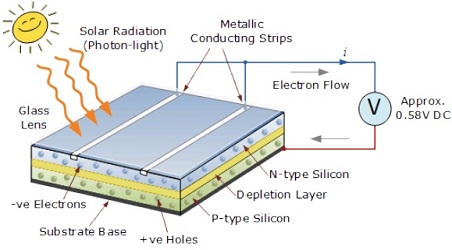
Mainly Solar cell is constructed using the crystalline Silicon that consists of a n-type semiconductor. This is the first or upper layer also known as emitter layer. The second layer is p-type semiconductor layer known as base layer. Both the layers are sandwiched and hence there is formation of p-n junction between them. The surface is coated with anti-reflection coating to avoid the loss of incident light energy due to reflection.
Working of Solar Cell
As soon as the solar cell is exposed to sunlight, the solar energy which is present in the form of light photons is absorbed by semi conductor materials. Due to this absorbed energy, the phenomena of photovoltaic occurs and electrons are liberated and produce the external DC current. The DC current is converted into 240-volt AC current using an inverter for different applications.
Photovoltaic cells are made of special materials called semiconductors such as silicon. An atom of silicon has 14 electrons, arranged in three different shells. The outer shell has 4 electrons. Therefore a silicon atom will always look for ways to fill up its last shell, and to do this, it will share electrons with four nearby atoms.
Now we use phosphorus (with 5 electrons in its outer shell). Therefore when it combines with silicon, one electron remains free. When energy is added to pure silicon it can cause few electrons to break free of their bonds and leave their atoms. These are called free carriers, which move randomly around the crystalline lattice looking for holes to fall into and carrying an electrical current.
However, they are very few and are not very useful. But impure silicon with phosphorous atoms takes a lot less energy to knock loose of “extra“ electrons because they are not tied up in a bond with any neighboring atoms. As a result, we have a lot more free carriers than we would have in pure silicon to become N-type silicon.
Check your knowledge of solar cells with our solar cell MCQs.
How Current Flows in Solar Cell
The other part of a solar cell is doped with the element boron (with 3 electrons in its outer shell) to become P-type silicon. Now, when this two type of silicon interact, an electric field forms at the junction which prevents more electrons to move to P-side. When photon hits solar cell, its solar energy breaks apart electron-hole pairs. Each photon with enough energy will normally free exactly one electron, resulting in a free hole as well. If this happens close enough to the electric field, this causes disruption of electrical neutrality, and if we provide an external current path, electrons will flow through the P side to unite with holes that the electric field sent there, doing work for us along the way. This electron flow provides the current.
Electron Hole Formation
As we know that photon is a flux of light particles and photovoltaic energy conversion relies on the number of photons striking the earth. On a clear day, about 4.4 x 1017 photons strike a square centimeter of the Earth’s surface every second. Only some of these photons that are having energy in excess of the band gap are convertible to electricity by the solar cell. When such photon enters the semiconductor, it may be absorbed and promote an electron from the valence band to the conduction band, that creates a hole in the valence band. After that the electron in the conduction band and hole in valence band combine together and forms electron-hole pairs.
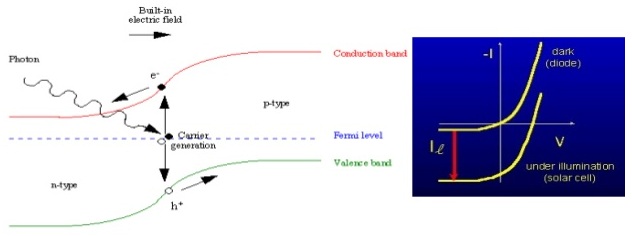
Thus when we connect these p and n layers to external circuit, electrons flow from n-layer to p-layer, hence current is generated. The electrons that leave the solar cell as current give up their energy to whatever is connected to the solar cell, and then re-enter the solar cell. Once back in the solar cell, the process begins again to produce more solar energy.
Types of Solar cell & Efficiency Levels
Depending upon the crystalline structure there are three types of solar cells:
- Mono crystalline silicon cells
- Polycrystalline silicon cells
- Amorphous silicon cells
The Mono crystalline silicon cell is produced from pure silicon (single crystal). Since the Mono crystalline silicon is pure and defect free, the efficiency of cell is higher. Efficiency of this type of solar cell is 14-17 %.
Polycrystalline solar cells use liquid silicon as raw material. Since the polycrystalline silicon involves solidification process the materials contain various crystalline sizes. Hence, the efficiency of this type of cell is less than Mono crystalline solar cell. Efficiency of this type of solar cell is 13-15 %.
Amorphous silicon cells are developed by depositing silicon film on the substrate like glass plate. The thickness of the layer is less than 1µm. Efficiency of this type of solar cells is 5-7 %.
Technology wise there are three types of solar cell technology:
- Discrete Cell technology,
- Integrated Thin Film technology and
- Multi crystalline Silicon technology.
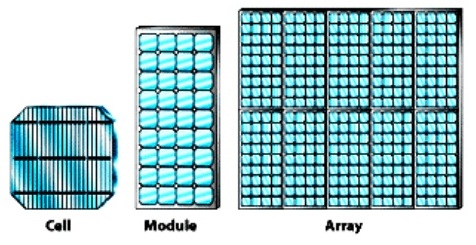
Solar panel (or) solar array (or) Solar module
The solar panel (or) solar array is the interconnection of number of solar module to get efficient power. A solar module consists of number of interconnected solar cells. These interconnected cells embedded between two glass plate to protect from the bad whether. Since absorption area of module is high, more energy can be produced.
Solar Cell Advantages
- Solar energy is clean and non-polluting.
- This is best renewable energy
- Solar cells do not produce noise
- Solar cell needs little maintenance
- Highly reliable and long lasting
- Operation costs are minimal
- Solar panel cost is now reducing
Discover more from Electrical Engineering 123
Subscribe to get the latest posts sent to your email.

One thought on “Solar Cell Construction & Working Principle”
Comments are closed.